
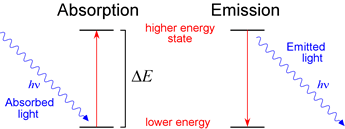
The gaseous layers of the Sun contains mainly isotopes of hydrogen and helium. Atoms within these layers will absorb light of specific frequencies/wavelengths to form an absorption spectrum. Reflected sunlight demonstrates absorption spectra of molecules in the atmosphere.īefore reaching Earth's surface, sunlight travels through gaseous layers of the Sun and Earth's atmosphere. Each emission line corresponds to the energy released for one electronic transition from excited to ground states. This produces an emission spectrum.ĭispersion of pink light from a hydrogen gas discharge tube produces an emission spectrum of multiple emission lines. When the light emitted from a gas discharge tube is dispersed through a glass prism, the light is split into its constituent wavelengths. This means the glass tube will radiate a particular colour. Afterwards, electrons will emit this energy in the form of electromagnetic radiation and return to the ground state.ĭepending on the identity of the gas, the emitted radiation may fall in the spectrum of visible light. If the energy is great enough, it will cause excite the electrons. When electrons in the current collide with gas molecules in the glass tube, their energy are partially transferred to the electrons in the gas atoms. Overall, reduced pressure facilitates conduction of current in a gas medium. Reduced movement leads to lower rate of molecular collision such that electrons’ motion (affected by the electric field) are less impeded.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
